Spotlight Fisher & Paykel Healthcare: A Pandemic Study in Embracing Change
June 26, 2020How does a Knowledge Leader handle a global pandemic? By adapting.
Consider New Zealand-based Fisher & Paykel Healthcare, a top 10 manufacturer in the global ventilator market, a company one could argue was already prepared for the unknown by its disciplined approach to investing in the future.
The firm’s long-term innovation footprint is easy to spot: since 2001, Fisher & Paykel has invested over $750 million cumulatively in R&D, a core value is “to be relentless in the pursuit of healthcare innovation,” and management commits 10% of each year’s revenue to R&D. As a result, according to our intangible-adjusted data Fisher & Paykel Healthcare is the No. 1 most innovative Health Care Equipment company in New Zealand and No. 3 in developed Asia, behind Cochlear Unlimited (Australia) and Olympus Corp (Japan), as measured by R&D as a percent of sales.
It was from this intellectual property-rich position that the company went about its business as usual at the end of 2019, celebrating a 50th anniversary milestone and hailing the recent launch of a new respiratory humidifier for intubated neonatal patients. To be clear, Fisher & Paykel does not manufacture the mechanical ventilators being used to treat COVID-19 patients in ICUs today around the world. Rather it manufactures the associated humidifiers and breathing circuits paired with ventilators that mimic the body’s natural humidification process to protect airway and lung function while a patient is on artificial breathing support. In 1971, Fisher & Paykel introduced the first respiratory humidifier based on a prototype made from a glass jar. The idea was to use warm, humidified air to maintain the natural balance of heat and moisture in the airways. Today the firm has extended that technology into product lines for the range of respiratory care. Sixty percent of revenues come from hospitals, for ventilation support, nasal oxygen delivery and surgical technology. The rest come from home care products, like CPAP machines for chronic conditions like sleep apnea and COPD.
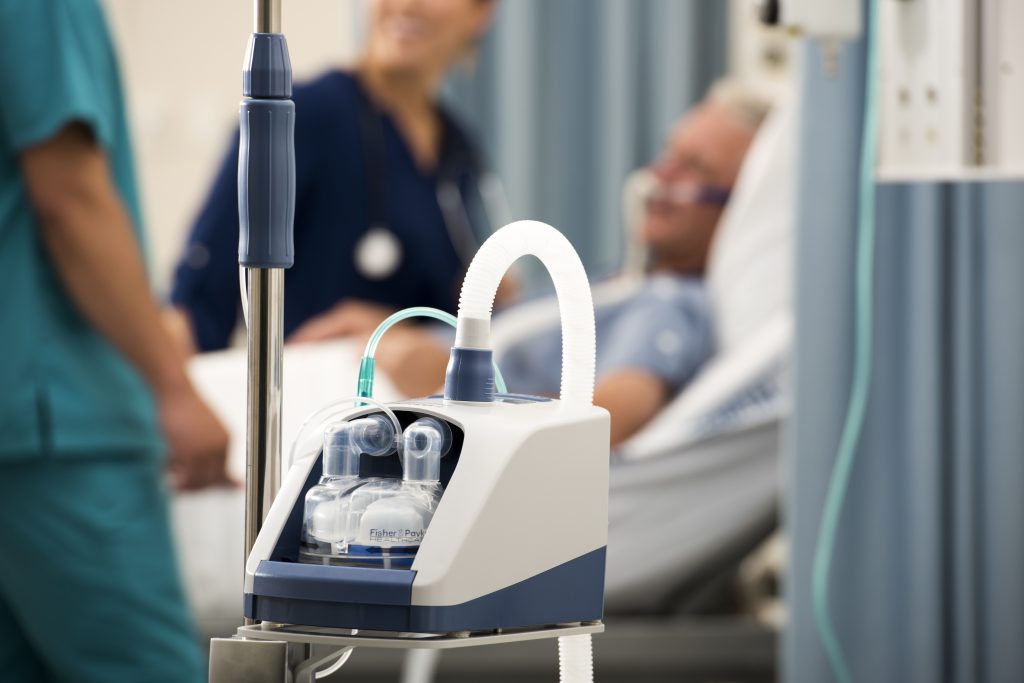
Airvo humidifier and integrated flow generator used in hospitals, credit: Fisher & Paykel Healthcare
In a sign of how fast times change, the firm released an interim annual report in November of 2019. Ironically, management’s major observation then was, “our results were also assisted by the extended flu season in the United States.” Major initiatives included the rollout of a new SAP enterprise database, the opening of a new manufacturing facility in Mexico and a new certification of its products for hospitals in Canada. In hindsight, each of these incremental results of the firm’s normal innovation engine can be viewed as investment in a future into which the company had no visibility.
There was still no indication a global pandemic was about to turn the firm’s operations upside down on February 3, 2020, when Fisher & Paykel announced a new nasal mask for sleep apnea: it would turn out to be the firm’s last non-COVID-19-related news. By February 21, the company announced it had ramped up efforts to support increased demand from China related to the outbreak. “Our people have been working long hours to ship products quickly, assemble them, and meet the need for training, particularly in Wuhan,” said CEO Lewis Gradon. On March 17, management updated company guidance. “Our respiratory humidifiers and consumables are directly involved in treating patients with coronavirus. We have seen an increase in demand globally and ramped up our manufacturing output … I also want to express my deep gratitude to our employees, who are working tirelessly to manufacture our products, support operations and supply and train end users,” the CEO said.
Less than two weeks later, on March 23, New Zealand’s prime minister shut down the country and designated Fisher & Paykel Healthcare an essential service. Gradon said, “As we advised last week, we have ramped up our manufacturing output and this will continue under the Level 4 alert status. As an essential service, we are continuing to focus on meeting the global demand for our respiratory products that are directly involved in treating patients with COVID-19.” Sending home all employees not directly critical to manufacturing and supply operations to work remotely, Graydon added, “this is a rapidly changing situation.”
In our research, a company’s allocation of resources tends to help reveal its innovation footprint. The next news from Fisher & Paykel came on April 6, when the company delayed its full-year earnings release from late May to the end of June. “At the present time, we have nearly 5,000 people around the world who are focused on meeting the increased demand for our respiratory products, which are being used in the treatment of patients with COVID-19,” said Chief Financial Officer Lyndal York. “This time of year our financial controllers in our global offices would usually be dedicated to completing year end reporting … many of these people are currently working remotely—assisting with customer enquiries, getting product into the hands of customers and providing operational support. One of our core beliefs is doing what is best for the patient, and it should come as no surprise that we think these activities should take priority.” Fisher & Paykel Healthcare is expected to announce results next Monday, June 29.
According to the company, its products improved health outcomes for about 14 million patients around the world in 2018 alone. Approximately 99% of revenue comes from outside New Zealand, with 47% of sales to North America, 29% to Europe and 19% to Asia Pacific. Products are manufactured in New Zealand and Mexico and sold in more than 120 countries worldwide. Of its nearly 5,000 employees worldwide, more than 550 are engineers, scientists and physiologists principally engaged in clinical research and R&D.
As of 3/31/20 Fisher & Paykel Healthcare and Olympus Corp were held in the Knowledge Leaders Strategy and Cochlear Unlimited was not. Intangible-adjusted data as of 6/22/20.